Formalising the induction of patient and public involvement contributors on trial oversight committees | Research Involvement and Engagement | Full Text

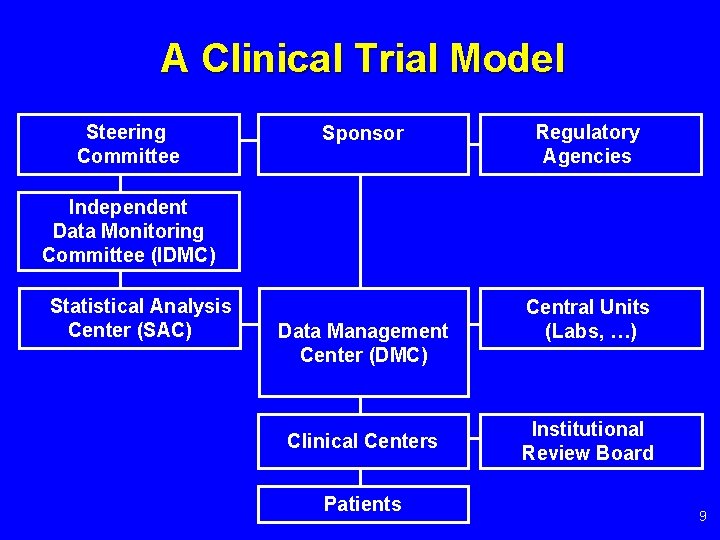
PPT - Independent Data Monitoring Committee (iDMC) and Role of A Biostatistician PowerPoint Presentation - ID:2295402

Importance and role of independent data monitoring committees (IDMCs) in oncology clinical trials | BMJ Open

Practical Issues in conducting multi-country studies by Ken Stanley, Ph.D. Harvard School of Public Health Collaboration Complexities 10 Basic Principles. - ppt download

Importance and role of independent data monitoring committees (IDMCs) in oncology clinical trials | BMJ Open
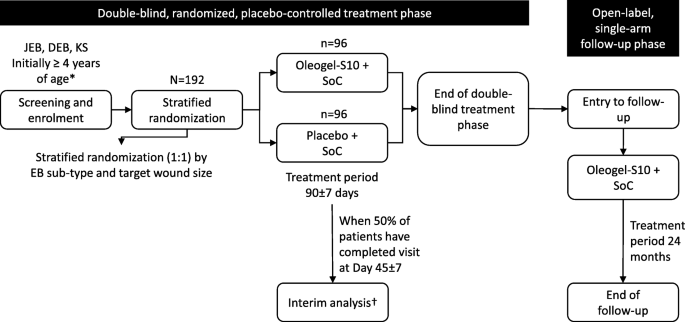
Oleogel-S10 Phase 3 study “EASE” for epidermolysis bullosa: study design and rationale | SpringerLink

Essential rules and requirements for global clinical trials in rare lung diseases: A sponsor׳s standpoint - ScienceDirect